Research Article
Tobi Victoria Omotoso*
Tobi Victoria Omotoso*
Corresponding Author
Department of Anatomy and Cell Biology, Obafemi
Awolowo University, Ile-Ife, Osun-State, Nigeria.
E-mail: omotosotobivictoria222@gmail.com, Tel: +23-08165563876
Victory Opeyemi Adekunle
Victory Opeyemi Adekunle
Department of Anatomy, University of Medical Sciences, Ondo State, Nigeria.
David Adesanya Ofusori
David Adesanya Ofusori
Department of Anatomy and Cell Biology, Obafemi Awolowo University, Ile-Ife, Osun-State, Nigeria.
Received: 2025-07-27 | Revised:2025-09-20 | Accepted: 2025-09-22 | Published: 2026-01-19
Pages: 01-10
DOI: https://doi.org/10.56717/jbt.2026.v02i01.06
Abstract
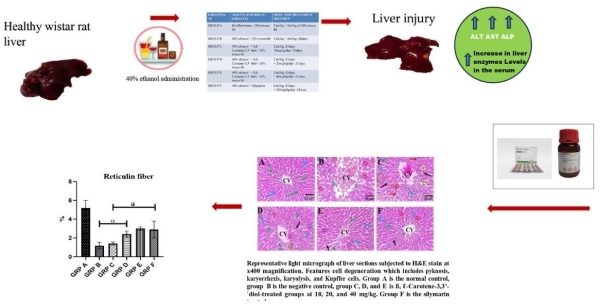
Liver injury is one of the most frequent life-threatening injuries in humans, caused by several factors such as viral agents, ethanol or drugs. The consumption of ethanol is globally, making it one of the leading risk factors for liver injury. This study aimed to evaluate the ameliorative effects of ß, Ɛ- carotene- 3, 3’-diol on ethanol-induced hepatotoxicity in rats. Forty-eight adult male Wistar rats (190 - 220 g) were randomly assigned into six groups (A-F) of eight rats each. Liver injury was induced in rats in groups B-F by the oral administration of 2 mL/kg b.w of 40% ethanol, once daily, for 21 days. After the last ethanol administration, the animals were fasted for twenty-four hours for gastric emptying. Thereafter, the animals in groups C-E were subjected to oral administration of ß, Ɛ-carotene-3,3’-diol, one dose every 12 h, for 21 days. Group F (positive control) rats were treated with oral administration of silymarin, at a dose of at a dose of 200 mg/bw in every 12 h, for 21 days. At the end of the experiment, the animals were sacrificed, the livers were excised and fixed for on histopathological and histomorphological analyses. The results showed that ethanol induced liver injury, characterized by the presence of pathological cell degeneration. Ethanol also reduced the number of intact hepatocytes and the percentage area of reticulin fibers. We conclude that treatment with ß, Ɛ-carotene-3,3’-diol mitigated ethanol-induced liver injury in a dose-dependent manner. This result highlights its ability to ameliorate ethanol-induced liver injury.
Abstract Keywords
Hepatotoxicity, ß, Ɛ-carotene-3,3’-diol, ethanol, pyknosis, karyorrhexis, karyolysis.
Reference
|
1. |
Gan,
C.; Yuan, Y.; Shen, H; et al. Liver diseases: epidemiology, causes, trends
and predictions. Signal Transduct. Target. Ther. 2025, 5,
33. https://doi.org/10.1038/s41392-024-02072-z. |
|
2. |
Coccolini,
F.; Coimbra, R.; Ordonez, C.; et al. M. Liver trauma: WSES 2020 guidelines. World J. of
Emerg. Med. 2020, 15, 1-15. https://doi.org/10.1186/s13017-020-00302-7 |
|
3. |
World
Health Organization. Global Status Report on Alcohol and Health. World
Health Organization: Geneva, Switzerland. 2018. ISBN 978-92-4-156563-9.
http://apps.who.int/iris. |
|
4. |
Wikipedia.
Ethanol history. https://en.wikipedia.org. Accessed: July, 2025. |
|
5. |
Alcohol
Use in the United States: Age Groups and Demographic Characteristics. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics-z/alcohol-facts-and-statistics/alcohol-use-united-states-age-groups-and-demographic-characteristics.
Accessed: August, 2025. |
|
6. |
Shield,
K.D; Parry, C; Rehm, J. Chronic diseases and conditions related to alcohol
use. Alcohol Res. 2014,
35 155-171. https://pubmed.ncbi.nlm,nih.gov/24881324/. Accessed:
June, 2025 |
|
7. |
Caputo,
F.; Guarino, M.; Casabianca, A; et al. Effects of Ethanol on the Digestive
System: A Narrative Review. J Trans.Gastro. 2024, 2, 186-192. https://doi.org/10.14218/JTG.2024.00028 |
|
8. |
Hyun,
J.; Han, J.; Lee, C; Yoon, M; Jung, Y. Pathophysiological Aspects of Alcohol
Metabolism in the Liver. Int. J. Mol. Sci. 2021, 22, 5717. https://doi.org/10.3390/ijms22115717. |
|
9. |
Cederbaum,
A.I. Alcohol metabolism. Clin. Liver Dis. 2012, 16, 667-685.
https://doi.org/10.1016/j.cld.2012.08.002 |
|
10. |
Devarbhavi,
H.; Asrani, S.; Arab, J.; Nartey, Y.; Pose, E.; Kamath, P. Global burden of
liver disease. J. Hepatol. 2023, 79, 516-537. https://doi.org/10.1016/j.jhep.2023.03.017 |
|
11. |
World
Health Organization. Global Status Report on Alcohol and Health. World
Health Organization. 2011. ISBN: 9789241564151. Accessed: February, 2025 |
|
12. |
Patel,
R.; Mueller, M. Alcoholic Liver Disease. 2023. |
|
13. |
Spearman,
C.W.; Sonderup, M.W. Health disparities in liver disease in Sub-saharan
Africa. Liver Int. 2015, 35, 2063-2071. https://doi.org/10.1111/liv.12884 |
|
14. |
Nwokediuko,
S.C.; Osuala, P.C; Uduma, U.V.; Alaneme, A.K.; Onwuka, C.C.; Mesigo, C.
Pattern of liver disease admissions in a Nigerian tertiary hospital.
Nig. J. Clin Pract. 2013, 16, 339-342. https://doi.org/10.4103/1119-3077.113458 |
|
15. |
Yan,
C.; Wanting, H.; Jinqi, T.; Jinyao, L.; Qionglin, L.; Shuxin, H. Pathogenic
mechanisms and regulatory factors involved in alcoholic liver
disease. J. Trans. Med. 2023, 21, 300. https://doi.org/10.1186/s12967-023-04166-8 |
|
16. |
Chen,
X. Protective effects of quercetin on liver injury induced by ethanol.
Pharmacogn. Mag. 2010, 6,
135-141. https://pmc.ncbi.nlm.nih.gov/articles/PMC2900062/ |
|
17. |
Namachivayam,
A.; Gopalakrishnan, A. Effect of Lauric acid against
ethanol-induced hepatotoxicity by modulating oxidative stress/apoptosis
signalling and HNF4α in Wistar albino rats. Heliyon. 2023, 9(e21267),
1-13. https://doi.org/10.1016/j.heliyon.2023.e21267 |
|
18. |
Francis,
P.; Navarro, V. Drug-Induced Hepatotoxicity. 2024.
https://pubmed.ncbi.nlm.nih.gov/32491467/ |
|
19. |
Brown,
S.; Mitchell, K.; Durning, M. Liver Disease: What You Should Know. Fatty
Liver Disease. 2025. https://www.webmd.com/fatty-liver-disease/liver-and-hepatic-diseases. |
|
20. |
Liu,
Y.L.; Reeves, H.L.; Burt, A.D.; Tiniakos, D.; McPherson, S.; Leathart, J.B.;
Anstee, Q.M. TM6SF2 rs58542926 influences hepatic fibrosis progression in
patients with non-alcoholic fatty liver disease. Nat. commun. 2014,
5, 4309. https://pubmed.ncbi.nlm.nih.gov/24978903/ |
|
21. |
Thurnham,
D.I.; Nolan, J.M.; Howard, A.N; Beatty, S. Macular response to
supplementation with differing xanthophyll formulations in subjects with and
without age-related macular degeneration. Graefes Arch. Clin. Exp.
Ophthalmol. 2015, 253,
1231-1243. https://pubmed.ncbi.nlm.nih.gov/25311651/ |
|
22. |
Sathasivam,
R.; Radhakrishnan, R.; Kim, J.; Park, S. An update on biosynthesis and
regulation of carotenoids in plants. South Afr. J. Bot. 2021, 140,
290-302. https://doi.org/10.1016/j.sajb.2020.05.015 |
|
23. |
Amang,
A.P.; Kodji, E.; Mezui, C.; Baane, M.P.; Siwe, G.T.; Kuissu, T.M.; Emakoua,
J.; Tan, P.V. Hepatoprotective Effects of Aqueous Extract of Opilia
celtidifolia (Opiliaceae) Leaves against Ethanol-Induced Liver Damage in
Rats. Evid. Based Complement. Alternat. Med. 2020, 1, 1-8.
https://doi.org/10.1155/2020/6297475 |
|
24. |
Song,
Z.; Deaciuc, I.; Song, M.; Lee, D.; Liu, Y.; Ji, X.; McClain, C. Silymarin
protects against acute ethanol-induced hepatotoxicity in mice. Alcohol. Clin.
Exp. Res. 2006, 30, 407–413. https://pubmed.ncbi.nlm.nih.gov/16499481/ |
|
25. |
Bruha,
R.; Dvorak, K.; Petrtyl, J. Alcoholic liver disease. World J. Hepat. 2012, 4,
81–90. https://pubmed.ncbi.nlm.nih.gov/22489260/ |
|
26. |
Scott,
S.; Kaner, E. Alcohol and public health: heavy drinking is a heavy price to
pay for populations. J. Public Health. 2014, 36, 396–398. https://pubmed.ncbi.nlm.nih.gov/25274327/ |
|
27. |
Hidayah,
D.R.; Triana, H.; Retno, M. Animal Models of Alcoholic Liver Disease for
Hepatoprotective Activity Evaluation. BIO Web of Conferences 2021, 41.
ISSN:2117-4458. https://wwww.bioconferences.org/articles/bioconf/abs/2021/13/bioconf_biomic2021_07007/bioconf_biomic2021-07007.html |
|
28. |
Sarpe
(Paduraru), A.M.; Dodul, C.; Vlase, E.A.; Onișor, C.; Niculet, E.; Ciobotaru,
O.C; Drima, E.P. A Review: Mental Manifestations and Biomarkers of Alcohol
Consumption. Life. 2024, 14, 873- 885.
https://doi.org/10.3390/ life14070873 |
|
29. |
|
|
30. |
Rojdmark.
S.; Calissendorff, J.; Brisma, K. Alcohol ingestion decreases both diurnal
and nocturnal secretion of leptin in healthy individuals. Clin. Endocrinol.
2001, 55, 639-647. https://pubmed.ncbi.nlm.nih.gov/11894976/ |
|
31. |
Adekunle,
V.O.; Ofusori, D.A. Morphometric Effects of ß, Ɛ-Carotene- 3,
3’-diol on Indomethacin- induced Gastric Ulcer in Adult Male Wistar Rats. J.
Anat. Sci. 2024, 15, 36-46. https://www.asn-ng.com/journal/article/1729770927 |
|
32. |
Chiang,
J. Liver physiology: Metabolism and Detoxifiction. Pathobiol. Hum. Dis.
2014, 11, 1770-1782. https://doi.org/10.1016/B9798-0-12-386456-7-04202-7 |
|
33. |
Boyd,
A.; Cain, O.; Chauhan, A.; Webb, G.J. Medical Liver Biopsy: Background,
Indications, Procedure and Histopathology. Front. Gastro. 2020, 11, 40–47. https://fg.bmj.com/content/flgastro/11/1/40.full.pdf |
|
34. |
Dasgupta,
A. Alcohol Biomarkers: An Overview. Clinical Aspects and Laboratory
Determination. ISBN 9780128003398. 2015;91-120.
https://www.sciencedirect.com/sciene/article/pii/B9780128003398000043 |
|
35. |
Sillanaukee,
P.; Massot, N.; Jousilahti, P.; Vartiainen. E.; Sundvall, J.; Olsson, U.;
Poikolainen, K.; Pönniö, M.; Allen, J.P.; Alho, H. Dose response of
laboratory markers to alcohol consumption in a general population. Am. J.
Epidem. 2000, 152, 747–751. https://pubmed.ncbi.nlm.nih.gov/11052552/ |
|
36. |
Conigrave,
K.; Davies, P.; Haber, P.; Whitfield, J. Traditional markers of excessive
alcohol use. Addiction. 2003, 98, 31–43. https://pubmed.ncbi.nlm.nih.gov/14984240/ |
|
37. |
Das,
M.; Basu, S.; Banerjee, B.; Sen, A.; Jana, K. Datta, G. Hepatoprotective
effects of green Capsicum annum against ethanol induced oxidative stress,
inflammation and apoptosis in rats. J. Ethnopharmacol. 2018, 227,
69–81. https://doi.org/10.1016/j.jep.2018.08.019 |
|
38. |
Wang,
X.; Dong, K.; Ma, Y.; Jin, Q.; Yin, S.; Wang, S. Hepatoprotective effects of
chamazulene against alcohol-induced liver damage by alleviation of oxidative
stress in rat models. Open Life Sci. 2020 15,
251–258. https://pubmed.ncbi.nlm.nih.gov/33817213/ |
|
39. |
Zatloukal,
K.; French, S.; et al. From Mallory to Mallory-Denk inclusion bodies: what,
how and why? Exp.
Cell Res. 2007, 313,
2033–2049. https://pubmed.ncbi.nlm.nih.gov/17531973/ |
|
40. |
|
|
41. |
Ofusori,
D.A.; Ofusori, A.E.; Awoniran J. Med. Plants. 2020, 14, 64-71.
doi:10.17311/rjmp.2020.64.71. |
|
42. |
Awoniran,
P.O.; Adeyemi, D.O. Ethanol extract of Curcuma longa rhizome
mitigates potassium bromate–induced liver changes in Wistar rats:
Histological, histochemical immunohistochemical assessments. Morphologie.
2018, 102, 276-288. https://doi.org/10.1016/j.morpho.2018.07.004. |
|
43. |
Ofusori,
A.E.; Raharjo, Y.; Ofusori, D.A.; Adekunle, V.O. Histomorphological effects
of ß, Ɛ-Carotene-3,3’-diol
on the liver following indomethacin-induced hepatotoxicity in adult male
Wistar rats. J. Med. Herbs Ethnomed. 2024, 10, 42-47.
https://doi.org/10.25081/jmhe.2024.v10.9014. |
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Liver injury is one of the most frequent life-threatening injuries in humans, caused by several factors such as viral agents, ethanol or drugs. The consumption of ethanol is globally, making it one of the leading risk factors for liver injury. This study aimed to evaluate the ameliorative effects of ß, Ɛ- carotene- 3, 3’-diol on ethanol-induced hepatotoxicity in rats. Forty-eight adult male Wistar rats (190 - 220 g) were randomly assigned into six groups (A-F) of eight rats each. Liver injury was induced in rats in groups B-F by the oral administration of 2 mL/kg b.w of 40% ethanol, once daily, for 21 days. After the last ethanol administration, the animals were fasted for twenty-four hours for gastric emptying. Thereafter, the animals in groups C-E were subjected to oral administration of ß, Ɛ-carotene-3,3’-diol, one dose every 12 h, for 21 days. Group F (positive control) rats were treated with oral administration of silymarin, at a dose of at a dose of 200 mg/bw in every 12 h, for 21 days. At the end of the experiment, the animals were sacrificed, the livers were excised and fixed for on histopathological and histomorphological analyses. The results showed that ethanol induced liver injury, characterized by the presence of pathological cell degeneration. Ethanol also reduced the number of intact hepatocytes and the percentage area of reticulin fibers. We conclude that treatment with ß, Ɛ-carotene-3,3’-diol mitigated ethanol-induced liver injury in a dose-dependent manner. This result highlights its ability to ameliorate ethanol-induced liver injury.
Abstract Keywords
Hepatotoxicity, ß, Ɛ-carotene-3,3’-diol, ethanol, pyknosis, karyorrhexis, karyolysis.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).

