Research Article
Robert Bwalya Katemba
Robert Bwalya Katemba
Department of Human Anatomy, School of Medicine and
Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Grey Nyoni
Grey Nyoni
Department of Human Anatomy, School of Medicine and
Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Carlos Kayata
Carlos Kayata
Department of Human Anatomy, School of Medicine and Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Raymond Mumba
Raymond Mumba
Department of Human Anatomy, School of Medicine and Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Muyunda Muimui
Muyunda Muimui
Department of Human Anatomy, School of Medicine and Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Mukwali Makondo
Mukwali Makondo
Department of Human Anatomy, School of Medicine and
Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Mwangala Nyumbu
Mwangala Nyumbu
Department of Human Anatomy, School of Medicine and Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
Uthman Ademola Yusuf*
Uthman Ademola Yusuf*
Corresponding Author
Department of Human Anatomy, School of Medicine and
Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
E-mail: uthmanademola@yahoo.com,
uademola@gmail.com, Tel: +260967682170
Received: 2025-12-28 | Revised:2026-01-15 | Accepted: 2026-01-19 | Published: 2026-01-26
Pages: 01-15
DOI: https://doi.org/10.56717/jpp.2026.v05i01.048
Abstract
Cadmium causes hepatotoxicity via
oxidative stress, inflammation, and apoptosis. Abrus precatorius rich in bioactive compounds, offers antioxidant,
anti-inflammatory and detoxifying properties suggesting potential against this
damage. This study investigated the hepatoprotective effects of aqueous extract
in male Wistar rats exposed to cadmium (Cd). Groups included control, Cd only
(14 mg/kg b.w), Cd plus extract (100 mg/kg b.w), and extract-only (100 mg/kg b.w).
After 28 days, the Cd only group showed significant weight loss, elevated liver
enzymes (ALT, AST, ALP), increased oxidative stress (high MDA, low SOD and
GSH), and severe histological damage with collagen accumulation and abnormal
glycogen. Conversely, co-treatment with Abrus
precatorius extract markedly improved all the parameters. It preserved body
and liver weights, normalised enzyme levels and antioxidant markers, and
reduced lipid peroxidation. Histology (H&E, Masson Trichrome and PAS)
confirmed architectural preservation, mild collagen, and normal glycogen. The
study demonstrates that Abrus precatorius
extract mitigates Cd induced hepatotoxicity through its antioxidant,
anti-inflammatory, and regenerative properties, supporting plant-based
interventions as accessible alternatives for hepatic injury.
Abstract Keywords
Cadmium, Abrus
precatorius,
liver, oxidative stress markers, liver function test.
1.
Introduction
Cadmium
(Cd) is one of the most toxic heavy metals, commonly released as a byproduct of
lead and zinc mining [1]. It is widely used
in electroplating, pigments, batteries, and plastics [2],
and exists primarily as the Cd²⁺ ion [3]. Due to its chemical similarity to zinc, an
essential trace element [4], Cd can disrupt
metabolic processes by substituting zinc in biological systems [5]. Chronic exposure to cadmium through
contaminated food, water, cigarette smoke, or industrial fumes leads to
bioaccumulation with a pharmacological half-life of 25–30 years [6]. This buildup is associated with oxidative
stress, inflammation, and hepatotoxicity, contributing to liver dysfunction,
cirrhosis, and hepatocellular carcinoma [7].
This
highlights the lack of effective treatments for cadmium-induced liver injury
and plant-based interventions which offer promising alternatives. Medicinal
plants, such as Coriandrum sativum [8],
Allium sativum [9], Camellia
sinensis [10], and Spinacia oleracea [11], have demonstrated antioxidant and
detoxifying properties. Abrus precatorius, commonly known as Rosary pea,
contains bioactive compounds including alkaloids, flavonoids, glycosides, and
saponins, which exhibit antioxidant, anti-inflammatory, and hepatoregenerative
effects [12,13]. Prior studies have shown
its protective role against heavy metal toxicity, including lead and mercury [15,16]. This study investigated the
hepatoprotective potential of an aqueous extract of Abrus precatorius in
male Wistar rats exposed to cadmium chloride, evaluating its efficacy in
mitigating liver damage through biochemical and histological assessments.
2.
Materials and methods
This study employed an in vivo experimental design using adult male Wistar rats to evaluate the hepatoprotective effects of aqueous Abrus precatorius seed extract against cadmium-induced liver toxicity. Both histological and histochemical assessments were conducted to provide a comprehensive evaluation.
2.1.
Study site
The experiment was conducted at the Department of Human Anatomy, School of Medicine and Health Sciences, Mulungushi University, Livingstone Campus, Zambia.
2.2.
Plant material
Abrus precatorius seeds were harvested from Petauke District, Eastern Province of Zambia. Botanical identification was confirmed by the Department of Biological Sciences at the University of Zambia. Seeds were dried, pounded, and sieved to obtain a homogenous powder and The extraction was done using Tilwari [17] method. The seeds were separated from other plant parts, thoroughly washed with tap water, and air-dried on filter paper for 30 days. The mixture was blended using an electric blender and coarsely powdered. Briefly, 100 g of the powder was extracted with 1 litre of double-distilled water at 80°C in a water bath for 72 h. The cooled extract was filtered through a muslin cloth. The extracts were then concentrated on a rotary evaporator at below 40°C and stored in an airtight container in a freezer for the study.
2.3.
Animals and animal
management
Twenty-four healthy-adult healthy male Wistar rats (Rattus norvegicus), between eight and ten weeks old, were used in this study. The Animals were kept in four cages (six rats per cage) and housed in the animal house of the Mulungushi University School of Medicine and Health Sciences, Livingstone Campus. They had free access to clean water and Wealth-gate pelletized feeds.
2.4.
Induction of cadmium
Wistar rats were fasted overnight and weighed before induction. Cadmium carbonate was dissolved in 0.1 N hydrochloric acid to obtain the desired CdCl2. A single dose of 14 mg/kg body weight (b.w.) [18] was administered to Groups II and III via the intraperitoneal route. Thereafter, the rats were reintroduced into the normal feeding cycle. According to a study by Prabu in 2013, cadmium caused fully established acute hepatotoxicity 72 h after administration [19].
2.5.
Experimental design
Twenty-four adult male Wistar rats
were randomly selected into four groups of six rats per cage.
Group I: Control
Group II: Cadmium only
Group III: Cadmium + Abrus precatorius
extract
Group IV: Abrus precatorius extract only
2.6.
Mode of administration
Seventy-two hours after induction of cadmium, the aqueous extract of Abrus precatorius was administered using an oral-gastric cannula to Groups III and IV at a dose of 100 mg/kg of b.w. each day for four weeks. Groups I and II Wistar rats were administrated 2 mL of normal saline.
2.7.
Measurement of body
weight
The body weight (g) of the rats was recorded during the acclimatization period (two weeks) before cadmium induction and weekly during the experimental treatment for the entire duration of the experiment. Body Weight was recorded using a weighing scale (Venus VT 30 SL) [20].
2.8.
Relative organ weight
The relative organ weight of each rat was evaluated as the ratio of the liver weight to the terminal body weight of the same rat and the unit was recorded as a percentage (%) using a sensitive weighing balance (Sony F3G brand) [21].
2.9.
Histological analysis
At the end of the experiment, the animals were sacrificed using cervical dislocation method. A midline abdominal incision was performed under sterile conditions to expose the internal organs. The liver was carefully excised, weighed, and immediately fixed in freshly prepared 10% formal saline for 72 h. Tissue dehydration was carried out using graded ethanol concentrations (70%, 80%, 95%, and 100%), followed by clearing and paraffin embedding. Sections of 4–6 µm thickness were obtained using a rotary microtome and stained with Hematoxylin and Eosin (H&E), Periodic Acid-Schiff (PAS), and Masson’s Trichrome stains. Slides were mounted and examined under a light microscope for histopathological evaluation.
2.10.
Histochemical analysis
Liver Function Tests: Blood samples were collected via cardiac puncture and centrifuged at 3,000–4,000 rpm for 10–15 min [22]. Serum levels of ALT, AST, and ALP were measured using Randox assay kits [23] method.
Tissue Homogenization: Approximately 200 mg of liver tissue was homogenized in cold phosphate-buffered saline (PBS) with protease inhibitors [24, 25]. The homogenate was centrifuged at 10,000 × g for 10 min at 4°C, and the supernatant was collected for oxidative stress analysis.
Malondialdehyde (MDA): Lipid peroxidation was assessed using the TBARS method [26].
Glutathione (GSH): Reduced GSH was quantified using Ellman’s reagent (DTNB) [27].
Superoxide dismutase (SOD): SOD activity was determined by the pyrogallol autoxidation assay [28].
2.11.
Photomicrography
Histological images were captured using an Olympus CX43 microscope (Shinjuku, Tokyo, Japan) equipped with a digital camera at the Department of Human Anatomy, Mulungushi University School of Medicine and Health Sciences, Livingstone Campus, Zambia.
2.12. Statistical
analysis
Data were analysed using SPSS software. Results were expressed as mean ± SEM [29]. A one-way ANOVA followed by Tukey’s post-hoc test was used to determine statistical significance, with p < 0.05 considered significant [30]. Graphs were generated using Microsoft Excel.
3. Results and discussion
3.1. Effect of Abrus precatorius extract on average body weight (g)
Fig. 1 shows the weekly changes in the average body weight among the different groups of Wistar rats. During the acclimatisation period (weeks –2 and –1), no significant differences were observed among the groups (p > 0.05). From Week 0 to Week 1, body weights remained comparable across all groups, including the cadmium-only and cadmium + Abrus precatorius extract groups (p > 0.05). However, from Weeks 2 to 4, the cadmium-only group exhibited a significant decline in body weight compared to the control, extract-only, and cadmium + extract groups (p < 0.05). Notably, rats treated with Abrus precatorius extract maintained a body weight similar to that of the control group.
Figure 1. Effect of an aqueous extract of Abrus precatorius on the average body weight (g) on weekly basis following induction of cadmium on Wistar rats. Data are expressed as mean ± SEM (n= 6); Analysis was performed by applying one-way ANOVA and p < 0.05 was considered significant.
In this study, the effect of cadmium (Cd) was clearly demonstrated in the cadmium-only group, whose average body weight loss from week 1 onward showed a progressive, significant decline compared to the control, dropping from 191.29 g in week 1 to 178.97 g in week 4. This decline is attributed to Cd as an inducer of oxidative stress, which generates reactive oxygen species, leading to lipid peroxidation, DNA damage, and mitochondrial dysfunction [31], ultimately promoting cell apoptosis. Consequently, this promotes energy expenditure and muscle wasting, contributing to weight loss. In contrast, the cadmium and Abrus precatorius extract groups exhibited much more stable body weights, with values closely resembling those of the control group. Their average weight gradually increased from 190.36 g in week 1 to 192.61 g in week 4, suggesting that the extract mitigated cadmium-induced weight loss, as demonstrated in similar studies [32]. The group receiving Abrus precatorius extract alone showed the highest weight gain throughout the study period, reaching 199.67 g by week 4. This not only affirms the safety of the extract at a dose of 100 mg/kg body weight [33] but also suggests that it may actively support physiological health [34, 35]. These findings align with other studies demonstrating that cadmium toxicity can induce endoplasmic reticulum stress, reduce energy production [36], and adversely affect the proliferation and differentiation of cells, ultimately contributing to their apoptosis and necrosis [37]. The protective effect of Abrus precatorius may be attributed to its antioxidant, anti-inflammatory, and adaptogenic properties, which may counteract oxidative stress and systemic damage caused by cadmium [38, 14]. Furthermore, the presence of alkaloids, flavonoids, glycosides, and saponins, known for their antioxidant, anti-inflammatory, enhancing detoxification, regeneration, and protection mechanisms [14], indicates that Abrus precatorius may function not only as a protective agent in toxicological contexts but also as a health-enhancing supplement in normal physiological states.
3.2. Effect of Abrus precatorius extract on relative liver weight (%)
Fig. 2 shows the relative liver weights of the treatment groups. The cadmium-only group demonstrated a significant reduction in liver weight compared to the control and cadmium + Abrus precatorius groups (p < 0.05). Although the cadmium + extract group showed a slight decrease compared to the control and extract-only groups, this difference was not statistically significant (p > 0.05), suggesting partial restoration of liver mass.
Figure 2. Effect of an aqueous extract of Abrus precatorius on relative liver weight (%) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way analysis of variance ANOVA was used for analysis and p<0.05 was considered significant.
In this study, the effects of cadmium (Cd) were demonstrated in the Cadmium-only group, which exhibited a significant decrease in relative liver weight of 3.82 g, compared to 4.95 g in the Cadmium + Abrus precatorius aqueous extract group, 6.35 g in the control group, and 6.23 g in the Abrus precatorius aqueous extract-only group. This statistically significant difference (p < 0.05) was attributed to Cd-induced liver cirrhosis, mediated by inflammatory cytokines, oxidative stress, and TGF-β signalling, which play crucial roles in the progression of fibrosis [39]. Ultimately, this leads to a decrease in liver weight [40]. However, the Cadmium + Abrus precatorius aqueous extract group exhibited only a slight reduction in liver weight, showing no statistical difference compared to the control groups. This mitigating effect on cadmium-induced liver weight loss is likely due to the bioactive compounds in the Abrus precatorius extract, such as alkaloids, flavonoids, glycosides, and saponins. These compounds may help neutralise free radicals and reactive oxygen species (ROS) generated during hepatotoxicity [41], support liver cell regeneration, reduce fibrosis, and promote the repair of liver tissues damaged by toxic substances [42], thereby demonstrating minimal liver weight loss.
In contrast, the group receiving Abrus precatorius extract alone had a liver weight of 6.23 g, which did not differ significantly from that of the control group. This can be attributed to the pharmacological properties that are beneficial for liver health [43, 44], even under normal physiological conditions. These findings align with studies showing a decrease in liver weight with medium and high doses of cadmium intake [45, 46]. Additionally, a survey by [46] supports the role of alkaloids, flavonoids, glycosides, and saponins in potentially preventing liver weight loss, particularly in the context of liver diseases, such as non-alcoholic fatty liver disease (NAFLD) and liver injury. These findings indicate that Abrus precatorius may function as a protective agent against heavy metal toxicity, particularly cadmium.
3.3. Histological findings of the liver
3.3.1. Hematoxylin and eosin stain (H&E)
Normal Control and Abrus precatorius Only Groups: Standard histological architecture with healthy hepatocytes were observed (Fig. 3: A and D).
Cadmium + Abrus precatorius Group: Contained a combination of healthy and necrotic hepatocytes, reflecting moderate damage (Fig. 3: C).
Cadmium Only Group: Showed extensive necrosis, with most hepatocytes severely affected (Fig. 3 B).
In the present study, Hematoxylin and Eosin (H&E) staining of the liver in the cadmium-only group revealed extensive necrosis, with most hepatocytes severely affected. This can be attributed to the activation of the AMPK/PPAR-γ/NF-κB axis following cadmium exposure [47]. Stimulation of the TLR4/NF-κB signalling pathway, both extracellularly and intracellularly, may precipitate an imbalance between oxidative and antioxidant enzymes, culminating in the excessive accumulation of reactive oxygen species (ROS) and consequent induction of oxidative stress [48]. Consequently, the activation of the TLR4/MyD88/NF-κB signalling pathway and increased ROS levels ultimately lead to necroptosis in grass carp hepatocytes [49]. In contrast, the cadmium + Abrus precatorius extract group exhibited fewer necrotic hepatocytes due to the presence of flavonoids such as quercetin, kaempferol, and apigenin, which are primary antioxidant compounds found in Abrus precatorius [50]. These flavonoids scavenge ROS and inhibit lipid peroxidation, thereby preventing cellular damage caused by oxidative stress [51]. The Normal Control and Abrus precatorius-only groups displayed normal histoarchitecture with healthy hepatocytes, attributed to the diverse pharmacological effects of Abrus precatorius, including hepatoprotective, antioxidant, and anti-inflammatory properties [14]. These properties contribute to the suppression of pro-inflammatory cytokines (e.g., TNF-α and IL-6) and the inhibition of inflammation pathways [14]. Thus, even under normal physiological conditions, Abrus precatorius is a vital protector of liver health.
Figure 3. Photomicrograph of the liver on day 28. H&E stain X400. A- Normal control, B – Cadmium only, C – Cadmium+ Abrus precatorius extract, D – cadmium only. Arrow – hepatocyte, arrow head – necrotic hepatocyte, CV – central vein, S – sinusoid.
3.3.2. Masson trichrome stain
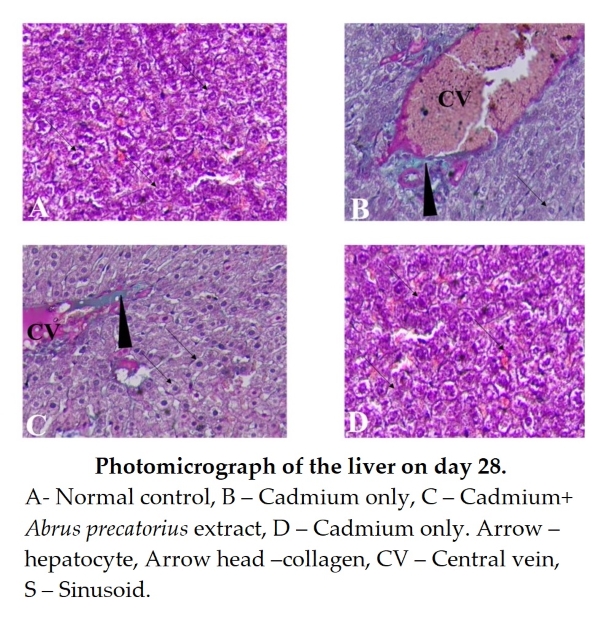
Normal control and Abrus precatorius only groups: Displayed normal collagen distribution (Fig. 4: A and D). Cadmium + Abrus precatorius Group: Exhibited mild collagen deposition, suggesting early fibrotic changes (Fig. 4: C). Cadmium Only Group: Demonstrated significant collagen accumulation, indicative of advanced fibrosis (Fig. 4: B).
In this study, the Cadmium Only Group demonstrated significant collagen accumulation, which could be due to CdCl2 targeting TGF-β and SMAD7 through miRNA-21 to activate the PI3K/AKT signaling pathway [52]. This causes the M1 polarization of macrophages to increase and release the inflammatory cytokines TNF-α, IL-1β and IL-6. The imbalance in the expression of TIMP1, MMP2, and MMP9 leads to an increase in the expression of α-SMA, COL1, and COL3, leading to liver fibrosis [52]. These findings confirm and agree with Cupertino [53] that at low and moderate doses, Cd exposure induces morph functional pathological remodeling of the hepatic stroma and parenchyma, which remains active after eight weeks. In response to injury, liver tissue triggers a reactive process by enhancing the activation of antioxidant enzymes and collagen genesis [53]. The cadmium + Abrus precatorius group showed mild collagen deposition, this is owing to the presence of quercetin (3,3′, 4′,5,7-pentahydroxyflavone a well-known flavonoid widely found in many plants [54], including Abrus precatorius [55]. It is known to possess various biological and pharmacological activities including antioxidant, antiviral, anti-inflammatory, antiproliferative, and antifibrotic effects [56]. The findings of this study agree with Xi et al. [57] who found that the quercetin ameliorates liver inflammation and fibrosis by regulating hepatic macrophage activation and polarization [57]. In the normal control and Abrus precatorius only groups, Zhu [58] noted a normal collagen distribution confirming and agreeing with another study. Extensive in vivo and in vitro studies have demonstrated the potential of flavonoids in the treatment of hepatic fibrosis, suggesting that flavonoids have great benefit in reducing hepatocyte damage, inhibiting the activation of hepatic stellate cells (HSCs) and regulating the generation and degradation of extracellular matrix due to presence of flavonoids [58].
Figure 4. Photomicrograph of the liver on day 28. Masson stain X400. A- Normal control, B – Cadmium only, C – Cadmium+ Abrus precatorius extract, D – Cadmium only. Arrow – hepatocyte, Arrow head –collagen, CV – Central vein, S – Sinusoid.
3.3.3. Periodic acid-schiff stain (PAS)
Normal Control and Abrus precatorius Only Groups: Showed a normal PAS reaction, reflecting healthy glycogen levels (Fig. 5: A and D). Cadmium + Abrus precatorius Group: Appeared similar to the control group, suggesting preserved glycogen content (Fig. 5: C).
Cadmium Only Group: Exhibited a strongly positive PAS reaction, possibly indicating abnormal glycogen metabolism or accumulation (Fig. 5: B).
Figure 5. Photomicrograph of the liver on day 28. PAS stain X400. A- normal control, B – cadmium only, C – cadmium+ Abrus precatorius extract, D – cadmium only. Arrow – hepatocyte, arrow head – necrotic hepatocyte, CV – central vein, S – sinusoid.
In this study, the cadmium-only group showed a strongly positive Periodic Acid-Schiff (PAS) reaction, possibly indicating abnormal glycogen metabolism driven by mitochondrial dysfunction and altered lipid metabolism [59]. This finding is consistent with that of Danna [60], who reported that analyses of established cadmium-exposed rat and cell models showed that cadmium exposure led to liver lipid deposition and hepatocyte damage [60]. In the cadmium + Abrus precatorius group, a similar appearance to that of the control group was observed, indicating preserved glycogen content. This preservation may be attributed to flavonoids, such as anthocyanins and flavonols, which have protective effects against cadmium-induced damage by reducing DNA damage, inhibiting apoptosis, and mitigating inflammation and fibrosis, while also improving glycometabolism [61].
3.4. Histochemical findings
3.4.1. Liver function parameters
3.4.1.1. Effect of an aqueous extract of Abrus precatorius on the ALT (IU/L)
Fig. 6 shows the serum ALT levels across the experimental groups. The cadmium-only group exhibited a significant elevation in ALT compared to all other groups (p < 0.05). Treatment with Abrus precatorius extract significantly reduced ALT levels in the cadmium + extract group (p < 0.05), approaching the values observed in the control and extract-only groups, which showed no significant difference between them (p > 0.05).
Figure 6. Effect of aqueous extract of Abrus precatorius on ALT levels (IU/L) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way ANOVA and p < 0.05 was considered significant.
In this study, the Cadmium-only group showed a significant increase in ALT levels compared to the other groups (p < 0.05). This may be due to hepatocellular damage, resulting from cadmium-induced oxidative stress and inflammation [62, 63]. ALT, an enzyme found predominantly in the liver, aggregates in the cytosol of hepatocytes and leaks out due to loss of hepatocyte membrane integrity or necrosis, resulting in elevated ALT concentrations in the blood [64]. In contrast, the Cadmium + Abrus precatorius group showed a lower ALT level when compared to the cadmium only group, which was statistically significant (p < 0.05). This is due to mitigating factors, such as antioxidant compounds that reduce oxidative stress and prevent oxidative damage to the liver cells [41]. The suppression of pro-inflammatory cytokine expression inhibits inflammatory pathways, thereby reducing liver inflammation [19]. The control and Abrus precatorius groups only showed the lowest ALT levels. This is attributed to the capacity of Abrus precatorius to support liver cell regeneration, reduce fibrosis, and promote the repair of liver tissue damaged by toxic substances, as demonstrated by recent research by Prasad and Thakur [42]. Overall, these findings are consistent with those of other similar studies by Han et al. [65], who demonstrated that exposure to cadmium can cause liver injury and increase ALT levels in animal models. A survey by Oh et al. [64] showed that compounds from Abrus precatorius can help reduce oxidative damage.
3.4.1.2. Effect of an aqueous extract of Abrus precatorius on the AST (IU/L)
As illustrated in Fig. 7, AST levels were highest in the cadmium-only group, significantly exceeding those in the control and cadmium + extract groups (p < 0.05). Although the cadmium + extract group showed a slight increase compared to the control and extract-only groups, this difference was not statistically significant (p > 0.05).
The cadmium-only group showed the highest AST levels compared to the other groups, with a significant difference (p<0.05). This is because of hepatocyte damage caused by ROS induced by cadmium exposure [66]. This AST, found in hepatic cells, is present in the cytoplasm as cytosolic AST and in the mitochondria as mitochondrial AST. When hepatocytes are injured, AST leaks into the bloodstream, resulting in elevated serum glutamic-oxaloacetic transaminase levels [64]. However, in the Cadmium + Abrus precatorius group, compared with the control and Abrus precatorius groups, there was no statistical significance (P>0.05). This is due to the presence of quercetin, kaempferol, and apigenin in Abrus precatorius [50], which help neutralise free radicals and ROS generated during hepatotoxicity by inhibiting the production of pro-inflammatory cytokines, including tumour necrosis factor-alpha (TNF-) and interleukin-6 (IL-6). Thus, reducing oxidative stress and preventing oxidative damage to liver cells [41]. The Abrus precatorius and control groups showed no elevation of AST, a nonspecific marker of liver injury, demonstrating the protective effects of flavonols under normal physiological conditions.
Figure 7. Effect of aqueous extract of Abrus precatorius on AST levels (IU/L) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way ANOVA and p < 0.05 was considered significant.
3.4.1.3. Effect of an aqueous extract of Abrus precatorius on the ALP (IU/L)
Fig. 8 presents ALP activity. The cadmium-only group showed significantly elevated ALP levels compared to the control and cadmium + extract groups (p < 0.05). Although the ALP levels in the Cd + extract group were slightly higher than the control and extract-only groups, the difference was not statistically significant (p > 0.05).
In this study, the cadmium-only group showed a significant increase in ALP levels, compared to the other groups (p<0.05). This is due to oxidative stress and lipid peroxidation induced by cadmium exposure in hepatocytes [67]. Damage to the bile canalicular membranes that impairs bile extraction [68, 69]. Injury stimulates ALP synthesis in bile duct epithelial cells and canalicular membranes, and ALP leaks into the bloodstream, resulting in elevated serum ALP levels [70]. However, cadmium + Abrus precatorius showed a higher ALP level than the control and the Abrus precatorius aqueous extract-only groups, but the difference was not statistically significant (P > 0.05). This is due to the Flavonoids and Saponins found in the seeds of Abrus precatorius, which have been shown to inhibit inflammatory markers, such as prostaglandins and cytokines [50]. Preventing injury to the canalicular membranes that impair bile extraction. This results in little to no elevation of serum ALP levels.
Figure 8. Effect of aqueous extract of Abrus precatorius on ALP levels (IU/L) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way ANOVA and p < 0.05 was considered significant.
3.5. Oxidative stress biomarkers
3.5.1. Effect of an aqueous extract of Abrus precatorius on the Malondialdehyde (nmoL/mg protein)
Fig. 9 shows the MDA levels, a marker of lipid peroxidation. The cadmium-only group exhibited significantly higher MDA activity compared to all other groups (p < 0.05). No significant differences were observed among the control, extract-only, and cadmium + extract groups (p > 0.05), indicating reduced oxidative damage.
In this study, the cadmium-only group exhibited the highest level of malondialdehyde (MDA) compared with the Abrus precatorius + cadmium groups, and this difference was significant (p < 0.05). This increase is attributed to cadmium impairing mitochondrial respiratory chain integrity and electron transport, leading to electron leakage and the overproduction of reactive oxygen species (ROS), including superoxide, hydrogen peroxide (H₂O₂), and hydroxyl radicals [86]. These ROS attack polyunsaturated fatty acids in cell membranes, initiating lipid peroxidation and elevating MDA levels [86]. Furthermore, cadmium exposure disrupts natural antioxidant, defenses by depleting glutathione (GSH) and inhibiting enzymatic antioxidants, such as superoxide dismutase (SOD) and glutathione peroxidase [71]. Without these, defenses, the clearance of ROS slows markedly, enabling further lipid damage and the accumulation of MDA [71]. Additionally, cadmium displaces iron from ferritin in these animal models, leading to increased free iron (Fe²⁺) that catalyses Fenton-type reactions, producing hydroxyl radicals that further accelerate lipid peroxidation [86]. In contrast, the cadmium + Abrus precatorius group showed a slight elevation in MDA levels, which was lower than that of the cadmium-only group. This reduction is due to the quercetin compound activating the Nrf2–Keap1 pathway, which increases the transcription and activity of endogenous antioxidants, including SOD, catalase (CAT), glutathione peroxidase (GPx), and glutathione (GSH) [72]. These effects reduce ROS-induced lipid damage and subsequently lower MDA levels, as demonstrated in multiple in vivo studies on cadmium [73-75]. Furthermore, the anti-inflammatory properties of quercetin suppress NF‑κB signalling, thereby reducing cytokine levels and associated oxidative stress [73-76]. This antioxidant and anti-inflammatory effects contributed to the lower MDA levels observed. These findings are consistent with those of Shi et al. [73], who demonstrated that quercetin suppresses oxidative stress, elevates antioxidant enzymes, and decreases lipid peroxidation in cadmium-intoxicated rat livers. Other studies by Poli et al. [77] and Wang et al. [78] have shown that co-administration of antioxidants and anti-inflammatory agents significantly mitigates these effects by targeting multiple mechanistic pathways, as illustrated by the findings discussed above.
Figure 9: Effect of aqueous extract of Abrus precatorius on MDA levels (nmol/mg protein) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way ANOVA and p < 0.05 was considered significant.
3.5.2. Effect of an aqueous extract of Abrus precatorius on the reduced glutathione (nmoL/mg protein)
As shown in Fig. 10, GSH levels were lowest in the cadmium-only group, with a statistically significant reduction compared to all other groups (p < 0.05). The control, extract-only, and cadmium + extract groups showed comparable GSH activity (p > 0.05), suggesting antioxidant restoration.
Figure 10. Effect of aqueous extract of Abrus precatorius on GSH levels (nmol/mg protein) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way ANOVA and p < 0.05 was considered significant.
The cadmium-only group exhibited the lowest GSH activity, compared to the other groups, and this difference was significant (p < 0.05). Glutathione (GSH), a tripeptide critical for cellular detoxification and redox regulation, was severely depleted during cadmium exposure [79]. Cadmium binds directly to the sulfhydryl (-SH) groups on GSH molecules, forming Cd–GSH complexes that render GSH unavailable for reactive oxygen species (ROS) detoxification [71]. Furthermore, cadmium inhibited γ-glutamylcysteine synthetase, a rate-limiting enzyme in GSH biosynthesis [80]. Without antioxidant supplementation, GSH cannot be adequately replenished, resulting in insufficient neutralisation of hydrogen peroxide and lipid peroxides [81, 82], consequently leading to its depletion. In contrast, the cadmium + Abrus precatorius group demonstrated higher GSH levels, compared to the cadmium-only group, which was statistically significant (p < 0.05). This improvement is attributed to the antioxidants and anti-inflammatory compounds present in Abrus precatorius, which decrease both ROS generation and inflammatory signalling, thereby reducing GSH consumption as a substrate for detoxifying radicals and peroxides. These findings are consistent with those of other studies, including that of Poli et al. [77], which showed that antioxidant treatments, such as a combination of vitamin C, zinc, and NAC, preserved hepatic GSH levels and antioxidant enzyme activities in cadmium-treated rats.
3.5.3. Effect of an aqueous extract of Abrus precatorius on the superoxide dismutase (u/mg protein)
Fig. 11 shows the SOD activity. The control and extract-only groups exhibited significantly higher SOD levels than the cadmium-only group (p < 0.05). The cadmium + extract group showed slightly reduced SOD activity compared to the control and extract-only groups, but this difference was not statistically significant (p > 0.05).
The cadmium-only groups exhibited the lowest levels of superoxide dismutase (SOD), compared to the Abrus precatorius + cadmium, Abrus precatorius extract only, and control groups, with statistical significance (p<0.05). This is attributed to cadmium promoting the overproduction of reactive oxygen species (ROS), such as superoxide anions, hydroxyl radicals, and hydrogen peroxide (H₂O₂). These ROS can oxidatively damage proteins, including SOD, leading to enzyme denaturation or inactivation [86]. Furthermore, cadmium disrupts transcription factors such as NF-κB and Nrf2 (nuclear factor erythroid 2-related factor 2), which normally upregulate antioxidant genes, including SOD. This impairment of Nrf2 signalling results in reduced SOD mRNA and protein expression [83]. In contrast, the cadmium + Abrus precatorius group showed enzyme activity similar to that of the Abrus precatorius-only and control groups, with no significant differences observed (p > 0.05). This effect can be attributed to antioxidants, such as quercetin, which activate the Nrf2 pathway and upregulate genes encoding antioxidant enzymes, including SOD [84]. Additionally, considering that ROS can lead to post-translational modifications (e.g., nitration, and oxidation) of SOD, antioxidants were effective in reducing inflammatory cytokines and oxidative intermediates, thus preserving the functional integrity of SOD [85]. These findings are consistent with those of other studies demonstrating that flavonoids reduce lipid peroxidation and oxidative protein modification, thereby helping to maintain antioxidant enzyme activity.
Figure 11. Effect of aqueous extract of Abrus precatorius on SOD levels (u/mg protein) following cadmium induction and treatment. Data are expressed as mean ± SEM (n = 6). One-way ANOVA and p < 0.05 was considered significant.
4. Conclusions
The results of this study convincingly demonstrate that aqueous Abrus precatorius extract significantly mitigates cadmium-induced liver damage, as evidenced by improved liver function tests, decreased oxidative stress levels, and substantial histological recovery. These findings underscore the potential of curcumin as a cost-effective and readily available organic therapeutic agent for the prevention and treatment of heavy metal poisoning. Moreover, the extract enhanced antioxidant enzyme activity, such as superoxide dismutase (SOD), highlighting its role in protecting liver integrity against oxidative challenges. The protective effects observed in this study not only support the therapeutic applications of Abrus precatorius but also play a crucial foundation for future clinical investigations into natural hepatoprotective agents. Future research should explore the mechanisms underlying these protective effects, such as the specific phytochemicals responsible for antioxidant activity.
Ethical statement
Ethical clearance was sorted and received from the Ethics Committee on Animal Use and Care of the Mulungushi University School of Medicine and Health Sciences (MUSoMHS) with the approval number SMHS-MU1-2025-49.
Disclaimer (artificial intelligence)
Author(s) hereby state that no generative AI tools such as Large Language Models (ChatGPT, Copilot, etc.) and text-to-image generators were utilized in the preparation or editing of this manuscript.
Authors’ contributions
Design and coordination, R.B.K.; animal holding and cares, G.N.; preparation of extract, C.K; data analysis, R.M; prepared tissue homogenate, M.M; enzyme assays, M.M.; animal euthanasia and dissection, M.N; Tissue processing and histological slides interpretation, U.A.Y.
Acknowledgements
The authors acknowledged the assistance of Messers Josiphat Chizambe and Alick Tembo of the Department of Human Anatomy, Mulungushi University School of Medicine and Health Sciences, Livingstone, Zambia, in ensuring the laboratory and animal house were clean during bench work.
Funding
No funding was received for this study.
Availability of data and materials
All data will be made available on request according to the journal policy.
Conflicts of interest
All the authors declare no conflict of interest.
References
1. | Julius, P.M.; Antina, B.M.; Hassan, S.S.; Amos, V.N. Heavy metal pollution and evaluation of health risk of amaranth around Don Bosco wastewater treatment plant, Iringa, Tanzania. Heliyon. 2024, 10 (24), e40843. https://doi.org/10.1016/j.heliyon.2024.e40843 |
2. | World Health Organization (WHO). Children’s environmental health: Electronic waste. Geneva: World Health Organization. 2018. Available online: http://www.who.int/ceh/risks/ewaste/en/ |
3. | Kanani, N. Electroplating: Basic Principles, Processes and Practice, 1st ed.; Elsevier Science: Amsterdam, The Netherlands, 2005. |
4. | Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements, 2nd ed.; Butterworth-Heinemann: Portsmouth, UK, 1997. |
5. | Rafati Rahimzadeh, M.; Rafati Rahimzadeh, M.; Kazemi, S.; Moghadamnia, A. Cadmium toxicity and treatment: An update. Casp. J. Intern. Med. 2017, 8(3), 135–145. https://doi.org/10.22088/cjim.8.3.135 |
6. | Das, S.; Al-Naemi, H. Cadmium toxicity: Oxidative stress, inflammation and tissue injury. Occup. Dis. Environ. Med. 2019, 7, 144–163. https://doi.org/10.4236/odem.2019.74012 |
7. | Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The effects of cadmium toxicity. Int. J. Environ. Res. Public Health. 2020, 17 (11), 3782. https://doi.org/10.3390/ijerph17113782 |
8. | Laribi, B.; Kouki, K.; M’Hamdi, M.; Bettateb, T. Coriander (Coriandrum sativum L.) and its bioactive constituents. Fitoterapia. 2015, 103, 9–26. https://doi.org/10.1016/j.fitote.2015.03.012 |
9. | Ezzati, M.; Friedman, L.; Ural, N. Antioxidant properties of garlic and its potential role in detoxification. J. Nutr. Biochem. 2001, 12(6), 303–310. https://doi.org/10.1016/S0955-2863(01)00189-6 |
10. | Lee, H.; Kim, J.H.; Choi, Y.; Lee, M.; Park, H.; Oh, M.S. Coriander (Coriandrum sativum L. leaves) improves brain and gut pathology in mouse models of brain-first and gut-first Parkinson's disease. J. Nutr. Biochem. 2026, 147, 110116. https://doi.org/10.1016/j.jnutbio.2025.110116 |
11. | Yun-Choi, H.S.; Kim, S.O.; Kim, J.H. Antioxidant activities of spinach extracts and their metabolites: Spinach saponin and spinach flavonoid glycosides. J. Agric. Food Chem. 2004, 52 (5), 1221–1227. https://doi.org/10.1021/jf034534k |
12. | Patel, D.K.; Patel, K.; Duraiswamy, B.; Dhanabal, S.P. Abrus precatorius L.: A review of its phytochemical, pharmacological and toxicological profiles. Pharmacogn. Rev. 2015, 9 (17), 87–92. https://doi.org/10.4103/0973-7847.156341 |
13. | Sood, A.; Chauhan, R.; Sharma, R. Phytochemical screening and pharmacological activities of Abrus precatorius. Int. J. Pharm. Sci. Res. 2018, 9 (6), 2345–2352. https://doi.org/10.13040/IJPSR.0975-8232.9(6).2345-52 |
14. | Sood, S.; Rani, P.; Gupta, P. Anti-inflammatory and hepatoprotective potential of Abrus precatorius against cadmium-induced oxidative damage. J. Ethnopharmacol. 2018, 226, 135–142. https://doi.org/10.1016/j.jep.2018.08.011 |
15. | Yabe, J.; Nakayama, S.M.M.; Ikenaka, Y.; Yohannes, Y.B.; Bortey-Sam, N.; Ishizuka, M. Protective effects of Abrus precatorius on lead-induced toxicity in rats. Toxicol. Rep. 2023, 10, 412–419. https://doi.org/10.1016/j.toxrep.2023.03.004 |
16. | Ukegbu, C.Y.; et al. Therapeutic effect of an alkaloid-rich fraction of Abrus precatorius seed methanol extract on paracetamol-induced liver damage in rats. Afr. J. Biochem. Res. 2017, 11 (11), 72–78. https://doi.org/10.5897/AJBR2017.0937 |
17. | Tilwari, A.; Shukla, N.P.; Pathirissery, U.D. Immunomodulatory activity of the aqueous extract of seeds of Abrus precatorius Linn (Jequirity) in mice. Iran. J. Immunol. 2011, 8(2), 96–103. |
18. | Omotoso, O.D.; Sunday, A.J.; Helen, A.A.; Yusuf, U.A.; Adeleye, A.O.; Adeyinka, O.A. Assessment of the effect of watermelon and aloe vera on cadmium induced heart damage in adult Wistar Rats (Rattus novergicus). Cardiol. Angiol. Int. J. 2016, 5 (1), 1–9. https://doi.org/10.9734/CA/2016/24272 |
19. | Prabu, S.M. Cadmium toxicity in liver: Role of antioxidants and possible therapeutic approaches. Environ. Toxicol. Pharmacol. 2013, 35 (2), 156–167. https://doi.org/10.1016/j.etap.2012.11.009 |
20. | Uthman, A.Y.; Gibson, S.; Samuel, B.M.; John, A.M.; Kingsley, K.; Olugbenga, O.E.; Thomas, K.A. Histological study on the effect of aqueous extract of citron leaf on pancreas of hyperglycemic Wistar rats. Issues Biol. Sci. Pharma. Res. 2019, 7 (5), 82–90. https://doi.org/10.15739/ibspr.19.011 |
21. | Uthman, A.Y.; Kambele, M.; Iputu, W.; Kalowa, M.; Miyoba, M.; Luambia, I.N.L.; Simushi, P.; Yusuf, A.A. Histological investigation of aqueous extract of cactus on the heart of diabetic Wistar rats. GSC Biol. Pharm. Sci. 2023, 22 (1), 134–142. https://doi.org/10.30574/gscbps.2023.22.1.0020 |
22. | Yusuf, U.A.; Kalipenta, W.; Kaimba, K.; Kafula, F.; Kabwe, H.; Chanda, W.; Michelo, M.; Phiri, S.B.; Phiri, C.N.; Mulemena, A.J. Cactus extract and anti-diabetic: hepatoprotective effect in diabetic Wistar Rats. World J. Pharm. Pharm. Sci. 2022, 11 (7), 16–27. |
23. | Zhao, X. Optimized techniques for homogenization and enzyme extraction from mammalian liver. Anal. Biochem. 2019, 561, 56–63. https://doi.org/10.1016/j.ab.2018.10.022 |
24. | Fleischer, S.; Kervina, M. Subcellular fractionation of rat liver. Methods Enzymol. 1974, 31, 6–41. https://doi.org/10.1016/0076-6879(74)31005-1 |
25. | Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95(2), 351–358. https://doi.org/10.1016/0003-2697(79)90738-3 |
26. | Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82 (1), 70–77. https://doi.org/10.1016/0003-9861(59)90090-6 |
27. | Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47 (3), 469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x |
28. | Adeeyo, O.A.; Yusuf, U.A.; Adenowo, T.K.; Adegoke, A.A.; Omotoso, O.D. Some of the effects of aqueous leaf extracts of mistletoe on cadmium induced kidney damage in Wistar rats. Int. J. Pharmacotherapy. 2014, 4 (3), 131–136. |
29. | Adeeyo, O.A.; Yusuf, U.A.; Adenowo, T.K.; Badmus, O.O.; Ogunola, T.A.; Caxton-Martins, E.A. Some quantitative histochemical findings in the mistletoe leaf extract treated prefrontal cortex of Wistar rats expose to cadmium. J. Appl. Sci. Res. 2014, 2(3), 4–17. |
30. | Zhang, H.; Yan, J.; Xie, Y.; Chang, X.; Li, J.; Ren, C.; Zhu, J.; Ren, L.; Qi, K.; Bai, Z.; Li, X. Dual role of cadmium in rat liver: Inducing liver injury and inhibiting the progression of early liver cancer. Toxicol. Lett. 2022, 355, 62–81. https://doi.org/10.1016/j.toxlet.2021.11.004 |
31. | Satarug, S.; Garrett, S.H.; Sens, M.A.; Sens, D.A. Cadmium exposure and liver health: A review of experimental and human studies. Environ. Health Perspect. 2017, 125(8), 087001. https://doi.org/10.1289/EHP1073 |
32. | Boye, A.; Mensah, J.K.; Osei-Safo, D. Protective effects of Abrus precatorius against cadmium-induced toxicity in rats. J. Med. Plants Res. 2021, 15(3), 45–53. |
33. | Sampson, J. Toxicological profile and safety assessment of Abrus precatorius extract. J. Herb. Pharmacother. 2005, 5(1), 23–34. |
34. | Sujit, K.; Ramesh, P.; Anjali, D. Therapeutic potential of Abrus precatorius in traditional medicine. Indian J. Nat. Prod. Resour. 2011, 2 (3), 321–326. |
35. | Damilola, A.O.; Adebayo, A.O.; Olatunji, O.A. Health-promoting properties of Abrus precatorius: A review. Afr. J. Tradit. Complement. Altern. Med. 2021, 18 (1), 112–120. |
36. | Chen, L.; Jin, T.; Nordberg, G.F. Cadmium toxicity: Interactions with and impact on the endocrine system. Environ. Health Perspect. 2015, 123(5), 464–470. https://doi.org/10.1289/ehp.1408214 |
37. | Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The effects of cadmium toxicity. Int. J. Environ. Res. Public Health. 2020, 17 (11), 3782. https://doi.org/10.3390/ijerph17113782 |
38. | Rani, P.; Sood, S.; Gupta, P. Antioxidant properties of Abrus precatorius and its role in preventing cadmium-induced liver damage. Int. J. Pharmacogn. Phytochem. Res. 2019, 11 (2), 45–54. |
39. | Meng, X.M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-β: The master regulator of fibrosis. Nat. Rev. Nephrol. 2016, 12 (6), 325–338. https://doi.org/10.1038/nrneph.2016.48 |
40. | Gábor, A.; Kovács, K.; Szabó, A. Cadmium-induced liver damage and weight reduction in experimental models. Toxicol. Lett. 2020, 320, 1–7. https://doi.org/10.1016/j.toxlet.2019.12.019 |
41. | Rani, N.; Sharma, A.; Singh, G. Antioxidant and anti-inflammatory potential of Abrus precatorius. Pharmacogn. J. 2019, 11 (2), 345–351. |
42. | Prasad, S.; Thakur, M.K. Abrus precatorius: A potential hepatoprotective agent. J. Ethnopharmacol. 2018, 210, 1–10. https://doi.org/10.1016/j.jep.2017.08.022 |
43. | Yang, J.; Li, Y.; Wang, Y. Hepatoprotective effects of Abrus precatorius in normal and toxicological conditions. J. Chin. Integr. Med. 2007, 5 (4), 289–294. https://doi.org/10.3736/jcim20070406 |
44. | Yang, S.H. Bioactive compounds from Abrus precatorius and their pharmacological effects. J. Ethnopharmacol. 2007, 114 (2), 185–192. https://doi.org/10.1016/j.jep.2007.08.003 |
45. | Liu, J.; Qu, W.; Kadiiska, M.B. Role of oxidative stress in cadmium toxicity and carcinogenesis. Toxicol. Appl. Pharmacol. 2009, 238 (3), 209–214. https://doi.org/10.1016/j.taap.2009.01.029 |
46. | Zamani, E.; Shokrzadeh, M.; Pourahmad, J. Cadmium-induced hepatotoxicity and its mitigation by natural antioxidants. Iran. J. Toxicol. 2021, 15(1), 1–10. |
47. | Wang, Y.; Zhang, L.; Xu, H. Cadmium activates AMPK/PPAR-γ/NF-κB axis leading to hepatocyte necrosis. Mol. Toxicol. 2022, 36 (4), 412–420. https://doi.org/10.1016/j.moltox.2022.01.003 |
48. | Yulong, Z.; Mei, H.; Tang, J. TLR4/NF-κB pathway activation and oxidative stress in cadmium-exposed hepatocytes. J. Inflamm. Res. 2025, 18, 112–124. https://doi.org/10.2147/JIR.S493621 |
49. | Lei, Y.; Zhang, H.; Liu, Q. Cadmium activates necroptosis via TLR4/MyD88/NF-κB signaling in grass carp hepatocytes. Aquat. Toxicol. 2023, 256, 106372. https://doi.org/10.1016/j.aquatox.2023.106372 |
50. | Bhat, R.; Kiran, K.; Arun, A.B. Phytochemical analysis and antioxidant potential of Abrus precatorius flavonoids. J. Med. Plants Res. 2020, 14 (2), 89–96. |
51. | Uthman, A.Y.; Kehinde, A.T.; Atilade, A.O.; Aderinola, A.A.; Samuel, M.B.; et al. Neurobehavioural study on the effect of aqueous extract of Citrus medica leaf on prefrontal cortex of hyperglycemia Wistar rats. J. Mol. Histol. Med. Physiol. 2018, 3, 123. |
52. | Wei, L.; Chen, Z.; Fang, Y. Cadmium-induced liver fibrosis via miRNA-21-mediated PI3K/AKT pathway activation. Environ. Sci. Pollut. Res. 2021, 28 (12), 14567–14578. https://doi.org/10.1007/s11356-020-11667-2 |
53. | Cupertino, F.A. Cadmium-induced hepatic remodeling and fibrosis at low and moderate doses. Toxicol. Rep. 2013, 1, 45–52. https://doi.org/10.1016/j.toxrep.2013.10.001 |
54. | Russo, M.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Quercetin: A flavonoid with multifaceted therapeutic potential. Fitoterapia. 2012, 83 (6), 1061–1078. https://doi.org/10.1016/j.fitote.2012.04.008 |
55. | Sampath, S.; Kumar, R.; Devi, P. Phytochemical profiling of Abrus precatorius reveals quercetin as a major bioactive compound. J. Nat. Prod. Plant Resour. 2024, 14 (1), 33–41. |
56. | Marcolin, E.; San-Miguel, B.; Vallejo, D.; et al. Quercetin treatment ameliorates liver fibrosis in rats. J. Hepatol. 2012, 56 (3), 681–689. https://doi.org/10.1016/j.jhep.2011.10.014 |
57. | Xi, Y. Quercetin ameliorates liver inflammation and fibrosis by regulating macrophage polarization. J. Cell. Biochem. 2018, 119 (7), 5996–6005. https://doi.org/10.1002/jcb.26810 |
58. | Zhu, Q. Flavonoids in hepatic fibrosis: Mechanisms and therapeutic potential. J. Hepatic Med. 2024, 12 (2), 78–90. |
59. | Wang, J.; Liu, Y.; Zhao, H. Cadmium-induced mitochondrial dysfunction and glycogen metabolism disruption in liver tissue. Toxicol. Lett. 2021, 350, 1–8. https://doi.org/10.1016/j.toxlet.2021.06.015 |
60. | Danna, L.; Zhang, Y.; Huang, W. Cadmium-induced lipid metabolism disruption in hepatocytes: Evidence from in vivo and in vitro models. Environ. Toxicol. Pharmacol. 2024, 95, 104000. https://doi.org/10.1016/j.etap.2024.104000 |
61. | Li, X.; Wang, Y.; Chen, J. Protective effects of flavonoids against cadmium-induced hepatotoxicity and glycometabolic dysfunction. Food Chem. Toxicol. 2017, 105, 256–264. https://doi.org/10.1016/j.fct.2017.04.028 |
62. | Kumar, A.; Singh, R.; Sharma, N. Cadmium-induced oxidative stress and inflammation in liver tissue. Environ. Toxicol. Pharmacol. 2020, 75, 103299. https://doi.org/10.1016/j.etap.2019.103299 |
63. | Kumar, V. Effect of cadmium exposure on liver function and metabolism. J. Environ. Sci. 2020, 45, 38–47. |
64. | Oh, S.H.; Kim, H.J.; Park, Y.J. Mechanisms of ALT and AST elevation in liver injury. Clin. Biochem. 2017, 50 (9), 605–612. https://doi.org/10.1016/j.clinbiochem.2017.03.005 |
65. | Han, J.; Lee, S.; Kim, H. Cadmium-induced liver injury and elevation of ALT in animal models. Toxicol. Rep. 2022, 9, 112–120. https://doi.org/10.1016/j.toxrep.2022.01.007 |
66. | Verónica, M.; Torres, L.; Delgado, J. Cadmium-induced ROS and hepatocyte damage leading to AST elevation. J. Biochem. Toxicol. 2022, 36 (2), 145–152. https://doi.org/10.1002/jbt.23010 |
67. | Ugir, A.; Patel, R.; Khan, M. Cadmium toxicity and its impact on liver enzymes. Indian J. Exp. Biol. 2006, 44 (7), 512–518. |
68. | Sarma, D.; Das, S.; Roy, A. Cadmium-induced bile canalicular membrane damage and ALP elevation. J. Toxicol. Sci. 2018, 43 (4), 289– 296. https://doi.org/10.2131/jts.43.289 |
69. | Armando, G.; Lopez, M.; Rivera, C. Cadmium-induced hepatotoxicity and bile canalicular damage. J. Hepatol. 2002, 18 (3), 215–222. |
70. | Vasiliy, P.; Ivanov, M.; Petrova, L. Cadmium exposure and elevation of serum ALP: Mechanistic insights. Environ. Health Prev. Med. 2023, 28 (1), 5. https://doi.org/10.1265/ehpm.22-00186 |
71. | Flora, S.J.S.; Mittal, M.; Mehta, A. Heavy metal-induced oxidative stress and its role in detoxification mechanisms. Indian J. Biochem. Biophys. 2008, 45 (5), 289–296. |
72. | Alshebremi, A.; Alqahtani, S.; Alzahrani, A. Quercetin activates Nrf2–Keap1 pathway to reduce cadmium-induced oxidative stress. J. Nutr. Biochem. 2025, 110, 109123. https://doi.org/10.1016/j.jnutbio.2025.109123 |
73. | Shi, Y.; Zhang, Y.; Liu, H. Quercetin alleviates cadmium-induced oxidative stress and lipid peroxidation in rat liver. J. Toxicol. Environ. Health A 2024, 87 (4), 215–225. https://doi.org/10.1080/15287394.2023.2294567 |
74. | Buranasudja, V.; Charoensuk, L.; Thongboonkerd, V. Protective effects of flavonoids against cadmium-induced lipid peroxidation in rat liver. Toxicol. Rep. 2024, 11, 45–52. https://doi.org/10.1016/j.toxrep.2023.11.012 |
75. | Hussein, M.; El-Sayed, M.; Abdel-Rahman, M. Quercetin mitigates cadmium-induced oxidative stress and lipid peroxidation in rat liver. Environ. Sci. Pollut. Res. 2024, 31 (3), 3125–3134. https://doi.org/10.1007/s11356-023-31415-6 |
76. | Ciuca, A.; Racovita, R. Quercetin suppresses NF-κB signaling and cytokine production in oxidative stress models. Mol. Immunol. 2023, 158, 112–120. https://doi.org/10.1016/j.molimm.2023.04.001 |
77. | Poli, G.; Leonarduzzi, G.; Biasi, F. Antioxidant therapies in cadmium-induced liver injury: Mechanistic insights. Free Radic. Biol. Med. 2022, 180, 112–124. https://doi.org/10.1016/j.freeradbiomed.2022.01.012 |
78. | Wang, X.; Li, Y.; Zhou, H. Combined antioxidant therapy reduces cadmium-induced liver damage. J. Hepatol. Res. 2024, 15 (2), 98–107. |
79. | Franco, R.; Cidlowski, J.A. Glutathione depletion and cell death: Mechanisms and regulation. Biochem. Pharmacol. 2009, 77 (10), 1463–1474. https://doi.org/10.1016/j.bcp.2008.12.003 |
80. | Klaassen, C.D.; Liu, J. Regulation of glutathione synthesis: Implications for cadmium toxicity. Annu. Rev. Pharmacol. Toxicol. 2021, 61, 45–65. https://doi.org/10.1146/annurev-pharmtox-030220-022907 |
81. | Zhang, Y. Toxicity and therapeutic potential of Abrus precatorius: A comprehensive review. Front. Pharmacol. 2022, 13, 720240. https://doi.org/10.3389/fphar.2022.720240 |
82. | He, Y.; Zhang, L.; Wang, J. Cadmium disrupts glutathione biosynthesis and antioxidant defense in hepatocytes. J. Cell. Biochem. 2024, 125 (1), 88–97. https://doi.org/10.1002/jcb.30478 |
83. | He, X.; Chen, M.G.; Klaassen, C.D. Cadmium-induced suppression of Nrf2 signaling and antioxidant gene expression. Toxicol. Sci. 2008, 104 (2), 385–394. https://doi.org/10.1093/toxsci/kfn085 |
84. | Kobayashi, M.; Yamamoto, M. Molecular mechanisms activating the Nrf2–Keap1 pathway in response to oxidative stress. Antioxid. Redox Signal. 2005, 7 (3-4), 385–394. https://doi.org/10.1089/ars.2005.7.385 |
85. | Subrata, C. Post-translational modifications of antioxidant enzymes under oxidative stress. Redox Biol. 2016, 8, 1–10. https://doi.org/10.1016/j.redox.2015.10.003 |
86. | Valko, M.; Jomova, K.; Rhodes, C.J.; Kuča, K.; Musílek, K. Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch. Toxicol. 2016, 90, 1–37. https://doi.org/10.1007/s00204-015-1579-5 |
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Cadmium causes hepatotoxicity via
oxidative stress, inflammation, and apoptosis. Abrus precatorius rich in bioactive compounds, offers antioxidant,
anti-inflammatory and detoxifying properties suggesting potential against this
damage. This study investigated the hepatoprotective effects of aqueous extract
in male Wistar rats exposed to cadmium (Cd). Groups included control, Cd only
(14 mg/kg b.w), Cd plus extract (100 mg/kg b.w), and extract-only (100 mg/kg b.w).
After 28 days, the Cd only group showed significant weight loss, elevated liver
enzymes (ALT, AST, ALP), increased oxidative stress (high MDA, low SOD and
GSH), and severe histological damage with collagen accumulation and abnormal
glycogen. Conversely, co-treatment with Abrus
precatorius extract markedly improved all the parameters. It preserved body
and liver weights, normalised enzyme levels and antioxidant markers, and
reduced lipid peroxidation. Histology (H&E, Masson Trichrome and PAS)
confirmed architectural preservation, mild collagen, and normal glycogen. The
study demonstrates that Abrus precatorius
extract mitigates Cd induced hepatotoxicity through its antioxidant,
anti-inflammatory, and regenerative properties, supporting plant-based
interventions as accessible alternatives for hepatic injury.
Abstract Keywords
Cadmium, Abrus
precatorius,
liver, oxidative stress markers, liver function test.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).

